
How to Improve Laboratory Turnaround Time
March 20, 2026A Startup Guide
From Regulatory Requirements to Revenue: Building In-House Drug Testing That Works
For pain management clinics, urine drug testing is no longer optional. It is embedded in the standard of care, required by state law in more than a dozen jurisdictions, and looked at by federal enforcement agencies whenever questions arise about opioid prescriptions. Every clinic that prescribes chronic opioids needs reliable, defensible toxicology data, and the question is no longer whether to test but how to structure testing in a way that supports patient care, satisfies payers, and withstands regulatory scrutiny.
For many clinics, the answer to the challenge is often testing in-house. Reference laboratory turnaround times stretch to days. Reimbursement for send-out testing often flows to the reference lab rather than the clinic. And regulatory pressure continues to push for individualized, risk-based testing, the kind a clinic can tailor to its patient population far more easily when it controls the testing workflow.
At Laboratory Management Consultants, we have guided pain management clinics across the country through the process of launching in-house toxicology programs. In this article, we’ll take a look at what it takes to build a compliant, profitable, and clinically sound drug testing operation. We’ll also cover the regulatory case for testing, CLIA certification pathways, equipment choices, reimbursement considerations, and specimen handling best practices.
The Clinical and Regulatory Case for In-House Toxicology
The American Society of Interventional Pain Physicians (ASIPP) published comprehensive evidence-based guidelines for opioid prescribing in chronic non-cancer pain in December 2023. These guidelines establish urine drug testing as a foundational element of responsible opioid management. UDT must be implemented from the initiation of chronic opioid therapy and continued as part of ongoing monitoring to reduce prescription drug abuse and illicit drug use.
ASIPP’s 2023 guidelines recommend that urine drug monitoring (UDM) be implemented at the initiation of chronic opioid therapy and conducted periodically for monitoring therapeutic compliance as per available guidance on mode and frequency of testing. Many Medicare Local Coverage Determinations and commercial payer policies translate this into the following common risk-stratified testing frequencies that pain clinics should consider when building protocols and documenting medical necessity:
- Low-risk patients: up to 2 presumptive and 2 definitive UDTs per rolling 365 days
- Moderate-risk patients: up to 2 presumptive and 2 definitive UDTs per rolling 180 days
- High-risk patients: up to 3 presumptive and 3 definitive UDTs per rolling 90 days
The CDC’s 2022 Clinical Practice Guideline for Prescribing Opioids for Pain, which replaced the 2016 guideline, softened its language slightly, recommending that clinicians consider the benefits and risks of toxicology testing for patients on subacute or chronic opioid therapy. Despite the softer language, federal enforcement trends show that urine drug test documentation remains a critical piece of evidence whenever there are opioid prescribing investigations. A 2022 content analysis in Pain Medicine identified 32 laws across 13 states that require UDT when opioids are prescribed for chronic pain, with frequency mandates varying by jurisdiction.
For pain clinics, the practical implication is clear: testing is not simply a clinical preference, it is a compliance expectation. The question becomes how to perform that testing efficiently, defensibly, and in a way that serves patients well.
In-house testing offers several advantages. Turnaround time drops from days to hours, or even minutes for point-of-care screening. Clinicians can act on results during the same visit when patterns warrant an immediate conversation. Revenue that would have gone to reference laboratories stays with the clinic. And the clinic can build workflows tailored to its specific patient population rather than adapting to a reference lab’s protocols.
CLIA Certification and Accreditation Pathways
Every laboratory that tests human specimens for diagnostic, treatment, or preventive purposes must operate under a CLIA certificate. The certificate type depends on the complexity of testing performed, and this is where many pain clinics make their first strategic decision.
Certificate of Waiver for CLIA-Waived Point-of-Care Testing
Many pain clinics begin with a Certificate of Waiver, which covers simple point-of-care urine drug test cups cleared by the FDA for waived testing. These cup-based immunoassay devices provide qualitative presumptive results within minutes and require minimal technical expertise. The Certificate of Waiver is inexpensive, quick to obtain, and requires laboratories to follow manufacturer instructions precisely.
Waived testing is a sensible entry point for low-volume clinics or those initially screening patients before referring to a higher-complexity laboratory for definitive confirmation. The tradeoff is limited sensitivity and specificity, along with a restricted test menu. Only FDA-cleared waived devices can be used.
Certificate of Compliance or Accreditation for Moderate-Complexity Testing
Clinics that want greater test menu flexibility, better screening capability, and higher throughput move to moderate-complexity testing under either a Certificate of Compliance (for CMS-inspected labs) or a Certificate of Accreditation (for labs accredited by COLA, CAP, or similar organizations).
Moderate-complexity testing covers instrument-based immunoassay platforms such as the Abbott ImmTox 270 and drugs-of-abuse panels run on clinical chemistry analyzers such as the Medica EasyRA. These systems deliver better sensitivity, broader drug class coverage, and the ability to run substantial daily volumes.
Personnel requirements are more demanding: moderate-complexity labs must have a qualified laboratory director, a technical consultant, and testing personnel meeting CLIA qualification standards.
COLA is particularly well-suited to pain management clinic laboratories because of its educational approach to accreditation and its focus on smaller clinical facilities. CAP accreditation is available for clinics seeking the gold standard, though it typically represents more overhead than most pain clinic labs require. Our Laboratory Setup & Accreditation Guide walks through the full decision framework for each pathway.
High-Complexity Testing for Definitive LC-MS/MS
Clinics adding LC-MS/MS confirmation capability operate under high-complexity rules. This opens access to the most sensitive and specific methods available for confirming and quantifying drugs and metabolites, but it also raises the bar for personnel qualifications. High-complexity laboratory directors must meet specific credentialing standards under CLIA, and technical supervisors must possess appropriate doctoral or master’s-level credentials with documented experience.
Because of the personnel requirements, many pain management clinic laboratories choose to partner with a part-time laboratory director through services like Laboratory Management Consultants rather than recruit a full-time director. This approach keeps costs manageable while maintaining regulatory compliance.
Equipment Selection for a Toxicology Startup
Equipment decisions drive everything else in a toxicology lab, including CLIA complexity level, personnel requirements, test menu, and reimbursement opportunities. Matching platform capability to clinical volume is the starting point.
Cup-Based Point-of-Care Immunoassay
CLIA-waived urine drug test cups are the simplest entry point. A patient voids directly into a cup with built-in reagent strips that produce a qualitative (presumptive) result in minutes. These are excellent for point-of-care screening and for settings where immediate results guide the clinical conversation. The limitation is sensitivity. Cup devices can miss synthetic opioids like fentanyl, certain benzodiazepines, and some semisynthetic opioids unless the specific panel is designed to include those assays.
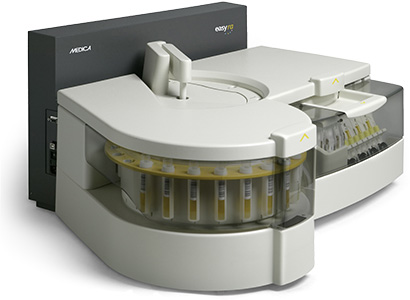
Medica EasyRA: Chemistry Analyzer with Drugs-of-Abuse Panel
For clinics that want to consolidate routine chemistry and toxicology screening on a single platform, the Medica EasyRA offers drugs-of-abuse urine screening alongside standard clinical chemistry. Throughput reaches 300 to 480 tests per hour depending on configuration, and the drugs-of-abuse assays operate under the same moderate-complexity CLIA classification as the chemistry panel. For a growing pain clinic that also performs basic metabolic testing in-house, the EasyRA delivers economic advantages through shared instrumentation. Our detailed article on the Medica EasyRA covers the platform in depth.
Abbott ImmTox 270: Dedicated Toxicology Immunoassay
When drug screening volume justifies dedicated equipment, the Abbott ImmTox 270 is purpose-built for the task. The benchtop analyzer processes up to 270 tests per hour, supports up to 24 unique assays in onboard storage, and handles batch processing for up to 30 patient samples at a time. For pain management clinics and addiction treatment centers processing 150 to 1,200 specimens monthly, the ImmTox 270 delivers efficiency without requiring complex infrastructure. Our Clinical Analyzer Buying Guide provides a full comparison of platforms, including the ImmTox 270.
Sciex LC-MS/MS for Definitive Confirmation
Presumptive immunoassay results require definitive confirmation whenever clinical decisions depend on the specificity of the finding. Immunoassays are susceptible to cross-reactivity and cannot reliably distinguish between structurally similar compounds. LC-MS/MS platforms such as the Sciex 3200 and 4500 provide the sensitivity, specificity, and quantitative accuracy that payers and regulators expect for confirmatory testing. These are high-complexity systems that require trained operators and ongoing maintenance, but for clinics aspiring to bring the full testing cycle in-house, LC-MS/MS is the gold standard.
Reimbursement Considerations
Understanding the coding structure for toxicology testing is essential to building a financially viable program. Payer policies are detailed and strictly enforced, and billing errors are a common source of claim denials.
Presumptive Testing CPT Codes
Presumptive drug testing uses one of three CPT codes based on testing methodology, and only one of these codes may be billed per patient per date of service:
- CPT 80305: Presumptive drug test capable of being read by direct optical observation only (dipsticks, cups, cards, cartridges). Includes specimen validation.
- CPT 80306: Presumptive drug test read by instrument-assisted direct optical observation (immunoassay using dipsticks, cups, cards, or cartridges). Includes specimen validation.
- CPT 80307: Presumptive drug test by instrument chemistry analyzers (immunoassay, chromatography, or mass spectrometry). Includes specimen validation.
Definitive Testing HCPCS Codes
For Medicare, definitive drug testing uses HCPCS codes rather than CPT codes 80320 through 80377. The G codes are tiered by the number of drug classes tested on a single specimen:
- G0480: 1 to 7 drug classes
- G0481: 8 to 14 drug classes
- G0482: 15 to 21 drug classes
- G0483: 22 or more drug classes
- G0659: Definitive testing performed at a less sophisticated level than typical reference laboratories
Only one definitive G code can be billed per date of service.
Common Billing Pitfalls
Several billing errors consistently trigger claim denials or audits:
- Specimen validity testing billed separately. Temperature, pH, specific gravity, and creatinine are included in the presumptive and definitive drug testing codes and cannot be billed as separate services.
- Standing orders without individualized medical necessity. Medicare and most commercial payers expect testing frequency to be documented as individualized to patient risk. Blanket testing protocols applied uniformly to all patients are predictable audit targets.
- Definitive testing without documented rationale. Definitive testing following a presumptive result requires documentation of why specificity is clinically necessary, such as an unexpected presumptive result, an adherence concern, or a safety evaluation.
- Using CPT 80320 through 80377 for Medicare definitive testing. These individual drug class codes are not accepted by Medicare for definitive testing. Use the G codes instead.
Risk-based testing frequency, aligned with ASIPP recommendations, is the strongest defensive posture against payer scrutiny. Documentation should connect each test to the patient’s current risk stratification and the clinical question the test will answer.
Specimen Collection and Chain-of-Custody Best Practices
Clinical toxicology testing in a pain management setting is not forensic testing, but the credibility of the results still depends on solid specimen collection practices. Regulators, payers, and surveyors all expect documented procedures that protect specimen integrity.
Temperature and Validity Checks
Federal guidelines specify that urine specimen temperature must be checked within four minutes of voiding and should fall between 90 degrees F and 100 degrees F (32 degrees C to 38 degrees C). Temperatures outside this range suggest possible substitution or tampering. Most collection cups include built-in temperature strips for immediate verification.
Additional validity markers include urine pH (normally 4.5 to 8), specific gravity (1.002 to 1.030), and creatinine concentration (greater than 20 mg/dL in normal urine). Results outside these ranges may indicate dilution, adulteration, or substitution. Clinics should have documented protocols for how to handle such findings.
Collection Environment
A properly designed collection facility controls obvious tampering opportunities:
- Taped or disabled faucets and bluing agent in the toilet water
- A secure area outside the restroom where patients leave bags, coats, and containers
- A collector who inspects the specimen for appearance and temperature within four minutes
- A minimum specimen volume of 30 mL for clinical testing (45 mL for federal workplace testing)
Handling Unexpected Results
One of the most common errors in clinical toxicology is acting on a presumptive result without confirmation. Immunoassays can produce false positives due to cross-reactivity, and false negatives for compounds the assay is not designed to detect. A positive presumptive result for a non-prescribed substance, or a negative presumptive result for a prescribed opioid, should trigger definitive confirmation before any clinical decision such as termination of therapy is made.
Pain management clinics should maintain written protocols for how clinicians respond to unexpected results, including patient conversation guidelines, confirmation testing pathways, and treatment plan adjustments. Document every step. An undocumented conversation cannot defend a decision months later if a regulator or attorney asks.
Documentation and Retention
Clinical drug testing does not require the formal federal chain-of-custody form used in workplace and legal testing, but documentation of collection, identity verification, specimen condition, and results handling should be consistent and retained. Most states and accrediting bodies require a two-year minimum record retention; some require longer. Verify your state requirements and build retention into your document control policy. Our Laboratory Staff Competency Assessment Guide covers the training documentation standards that support a defensible program.
How Laboratory Management Consultants Supports Toxicology Startups
Building a compliant, profitable toxicology laboratory in a pain management clinic setting requires coordination across regulatory, operational, technical, and financial domains. LMC integrates all of these into a single consulting engagement.
Our toxicology startup support includes:
- CLIA application guidance for Certificate of Waiver, Compliance, or Accreditation, with the paperwork and timeline management that keeps applications moving forward
- Equipment selection consulting that matches platform capability to clinical volume, physical space, and budget, drawing on our active buying-and-selling experience in the laboratory equipment market
- Part-time laboratory director services for moderate and high-complexity laboratories, meeting CLIA director qualification requirements without the cost of a full-time hire
- Customized policy and procedure manual development covering toxicology workflows, specimen collection, chain of custody, validity testing, and corrective action
- Validation study support for new instruments and methods, including precision, accuracy, linearity, and comparison-of-methods studies
- Accreditation preparation with mock surveys and ongoing compliance monitoring for COLA, CAP, or CMS inspections
- Reimbursement and coding consultation to help clinics build billing workflows that align with Medicare and commercial payer expectations
Frequently Asked Questions
Is urine drug testing required for patients on chronic opioid therapy?
ASIPP’s 2023 guidelines state that UDT must be implemented from the initiation of chronic opioid therapy and continued as part of ongoing adherence monitoring. The CDC’s 2022 guideline recommends that clinicians consider the benefits and risks of toxicology testing.
As of the 2022 content analysis published in Pain Medicine, 13 states had enacted laws requiring UDT when opioids are prescribed for chronic pain (with frequency mandates varying by jurisdiction); the number of states with such requirements has grown since that analysis.
What is the difference between presumptive and definitive drug testing?
Presumptive testing uses immunoassay or similar rapid methods to screen for the presence of drug classes. It is fast and economical but can produce false positives due to cross-reactivity and false negatives for compounds outside the assay’s target range. Definitive testing uses mass spectrometry methods (GC-MS or LC-MS/MS) to identify specific drugs and metabolites with high accuracy. Definitive testing is typically ordered to confirm unexpected presumptive results or when clinical decisions require precise identification.
What CLIA certificate does my pain clinic need for in-house drug testing?
A Certificate of Waiver covers basic point-of-care cup-based immunoassay testing. A Certificate of Compliance or Accreditation covers moderate-complexity instrument-based immunoassay testing on platforms such as the Abbott ImmTox 270 or the Medica EasyRA. High-complexity certification is required for LC-MS/MS definitive testing. The right choice depends on your expected volume, test menu, and staffing plan.
Can I bill for specimen validity testing separately?
No. Specimen validity testing (temperature, pH, specific gravity, creatinine) is included in the presumptive and definitive drug testing CPT and HCPCS code descriptions. Billing it separately under codes such as 81000, 81001, 81002, 82570, or 83986 will be denied and may trigger audit scrutiny.
How does Laboratory Management Consultants support new toxicology laboratories?
LMC provides integrated support across the full startup cycle: CLIA application guidance, equipment selection and validation, part-time laboratory director staffing, customized policy and procedure manuals, accreditation preparation, and reimbursement coding consultation. Our team has guided pain management clinics, addiction treatment centers, and physician office laboratories through this process across the country.
Launch Your Toxicology Program on Solid Ground
Pain management clinics face growing pressure to deliver rapid, defensible, cost-effective drug testing as part of responsible opioid prescribing. The clinics that build this capability in-house, with the right equipment and the right compliance structure, gain clinical speed, revenue, and regulatory confidence.
Contact Laboratory Management Consultants today at (606) 487-0523 or email tpace@emaillmc.com for a free toxicology startup consultation.
Visit us online at https://laboratorymanagementconsultants.org to learn more about our comprehensive laboratory support services.
Do not let regulatory complexity or equipment confusion delay your clinic’s toxicology program. With the right guidance, your laboratory can launch on solid ground and operate at peak compliance from day one.
Related Resources from LMC:











Medica EasyRA

Thermo Fisher Indiko Plus

Abbott ImmTox 270

The 3200 QTRAP system

The Pictus 500